 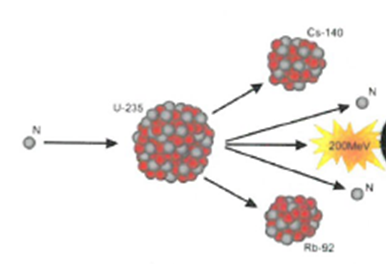
Nuclear fusion occurs in elements with a low atomic number, such as hydrogen, and is a highly reactive process. When two or more light nuclei collide, they fuse to form a heavier nucleus, which is known as nuclear fusion. Nuclear Fusion is the process of combining nuclear fuel with other elements to form a new fuel source. A single gramme of matter can release approximately 90,000,00000 kilojoules of energy, which is a huge amount of energy. In order to make up for this missing’ mass, it is converted into energy. To make things easier to understand, the products formed in nuclear fission and nuclear fusion always have a lower mass than the reactants in both processes. 
It is possible to define nuclear binding energy as the amount of energy required to keep all of the protons and neutrons contained within the nucleus.Ī nuclear reaction (such as a fission or fusion reaction) releases the mass accounted for by the nuclear binding energy according to the equation e = mc 2 (energy equals mass times the square of the speed of light), which is defined as mass times the square of the speed of light. This difference in mass is attributed to nuclear binding energy, which is a nuclear reaction (often referred to as a mass defect). Unlike other atomic nuclei, the mass of an atomic nucleus is always less than the sum of the individual masses of each subatomic particle that makes it up (protons and neutrons). Reactions in nuclear reactors release enormous amounts of energy The only thing that has happened is a transfer of momentum and energy. Nuclear Scattering Process: This process involves the collision and separation of atomic nuclei without altering the composition of the nucleus.Spontaneous Fission Reactions: These are reactions that take place without the presence of neutrons, and as a result, do not require the presence of neutrons to take place.The sole purpose of doing so is to deplete one’s energy reserves. Nuclear decay is a process in which an unstable nucleus tends to emit radiation as a result of its instability.However, unlike a Nuclear Reaction, these occur spontaneously rather than being induced. These processes appear to be similar to a Nuclear Reaction in their appearance. Specific processes that do not meet the definition of Nuclear Reactions are listed below. Nuclear Reactions are defined as external changes that occur on Atomic Nuclei as a result of their internal changes. Nuclear Fusion, on the other hand, refers to a process in which two light nuclei collide to form a single heavy nucleus as a result of the collision. Furthermore, it disintegrates into two or lighter Nuclei. The heavy nucleus in nuclear fission has a tendency to absorb neutrons or other relatively lighter particles. Nuclear Fission and Nuclear Fusion Reactions are the two types of nuclear reactions that are most commonly encountered. The Nuclides produced by Nuclear Reactions are not the same as the Nuclides produced by reacting nuclei or the parent nuclei. One or more nuclides are produced as a result of the collision of an Atomic Nucleus with another Atomic Nucleus or as a result of the collision of one Atomic Nucleus with a Subatomic Particle. Discover what a Nuclear Reaction is, what types of Nuclear Reactions there are, and some insightful frequently asked questions in this informative article. Nuclear decays, on the other hand, can be represented in a similar manner. Balanced Analogously, you can represent the Nuclear Reaction in an Equation that is similar to a Chemical Equation. If the Particles collide with each other without undergoing any change, the process is referred to as an Elastic Collision rather than a Reaction, and the process is classified as such. Also keep in mind that the Reaction can involve more than two colliding Particles, but this is a rare occurrence. If two nuclei or nuclear particles collide and produce products that are different from the original nuclei or nuclear particles, this is referred to as a Nuclear Reaction in nuclear chemistry and physics. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
